By their very nature, every permanent magnet contains some form of iron. Iron displays the most dramatic ferromagnetic properties of all elements, which is why it is found in the most powerful magnets. However, iron is highly reactive with water and then causes magnets with high iron content very susceptible to corrosion. Other ferromagnetic elements used in different types of permanent magnets include nickel, cobalt, gadolinium, and dysprosium, all of which have an impact on both magnetic strength and resistance to corrosion.
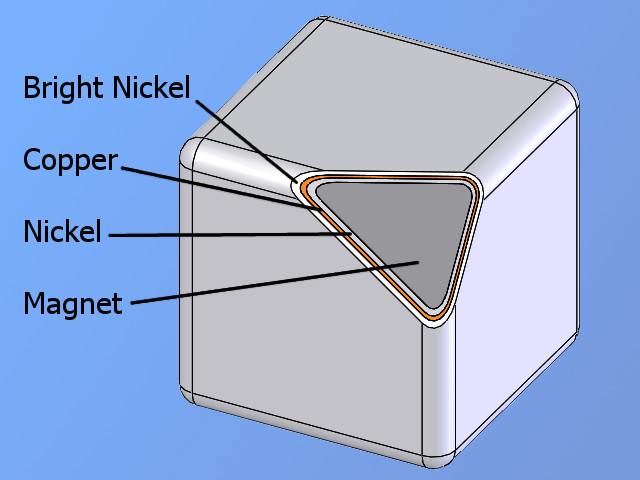
The strongest permanent magnets available are Neodymium magnets, but due to their high iron content (64-68%), they are also highly susceptible to corrosion in damp environments. Modern manufacturing techniques have seen traces of other rare earth elements, such as dysprosium added to the NdFeB alloy to improve resistance to corrosion as well as other magnetic properties. Because of their susceptibility to corrosion, Neodymium magnets are almost always coated before use. A very common coating used for permanent magnets is a triple layer of nickel-copper-nickel, however, there are still many other suitable options including but not limited to gold, silver, zinc, tin, epoxy, Teflon, and various paints and lacquers.

Samarium cobalt magnets, which like Neodymium magnets are part of the rare earth family, are much more resistant to corrosion even when uncoated. This is because much of the iron used in Neodymium magnets is replaced with cobalt, which does not react to water. You will still find many Samarium Cobalt magnets coated as standard but this is essentially for aesthetic purposes or for application-specific reasons such as adhesion or sterility. Samarium Cobalt magnets are widely used in marine and offshore applications when they have to cope with being constantly exposed to saltwater air.
Alnico and Ferrite magnets are both known for their resistance to corrosion, although they don’t provide the same level of magnetic performance as rare earth Samarium Cobalt and Neodymium magnets. Some grades of Alnico magnets do contain traces of iron and therefore will display some light surface corrosion over time if exposed to water. However, if exposed to other lubricants such as oil, solvents, or alcohol, Alnico performs very, very well. Sometimes, Alnico magnets have been coated in red paint; it is not clear how or why this custom started but it is one still followed today with most Alnico magnets instantly recognizable by their trademark red finish. Ferrite magnets, also known as Ceramic magnets, are made from iron oxide, and as a result, they simply do not corrode even when submerged in water. Because of their unrivaled resistance to corrosion, Ferrite magnets are very rarely coated, but as Ferrite is a dusty material some applications will require some surface treatment to avoid the dust spreading.




Nice post. I was checking constantly this weblog and I am
impressed! Extremely helpful info specially the last section 🙂 I maintain such information much.
I was seeking this particular info for a long time.
Thanks and best of luck.
Thanks for your comments, if you have any questions about magnet, feel free to contact us~